How do top performing companies plan and deliver profitable portfolios of smarter, more connected products? Join this webcast to hear what recent survey results tell us about developing innovative products in the digital age. The webcast includes results from the 6th Product Portfolio Management Benchmark study, conducted by Tech-Clarity and sponsored by Planview. Hear Tech-Clarity’s Jim…
- Include all functional departments early in the NPD process, for example inviting corporate IT to specify cloud and enterprise system enhancements
- Change the product design process to incorporate new decisions such as where to place sensors
- Adapt the product innovation process to take advantage of new insights available from smart, connected products in the field, including actual operating conditions
Conclusions and Recommendations
It’s time for companies to make the transition to designing smart, connected products. The business benefits are compelling, and we believe that those that implement these capabilities first will put their competitors at a disadvantage. To adapt, companies need to make changes to their NPD processes and technology. They need to involve more disciplines early in the design process, include a new series of decisions in their design process, and learn from real product performance by “closing the loop” between design and operation.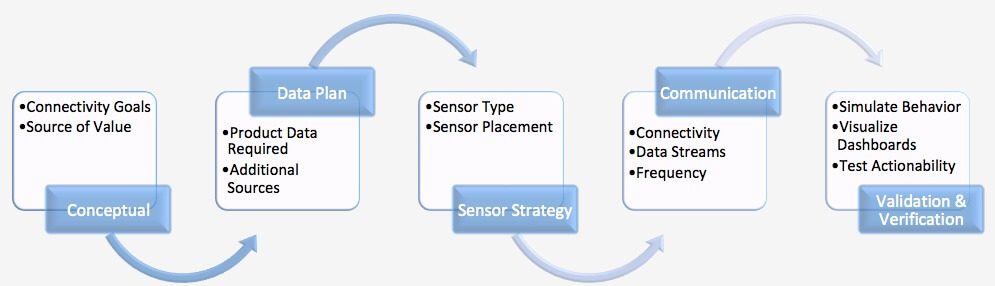 In order to take advantage of the digital opportunities, companies must evolve modeling, design collaboration, and simulation to support connected design including design decisions related to:
In order to take advantage of the digital opportunities, companies must evolve modeling, design collaboration, and simulation to support connected design including design decisions related to:
- Conceptual design and requirements
- Data design
- Sensor strategy
- Communication plan
- Validation and verification
It's time to act. With the move toward a sharing economy, we believe that companies that simply develop and sell products will put themselves at a significant disadvantage to companies that design, develop, and stay in touch with their products throughout the product’s life. Companies that don’t embrace digitalization and start offering more than a physical product stand the risk of being commoditized.
*This summary is an abbreviated version of the report and does not contain the full content. A link to download the full report is available above. If you have difficulty obtaining a copy of the report, please contact us using the "Contact" link below. [post_title] => Guide to Selecting a Smart, Connected Product Design Solution (eBook) [post_excerpt] => [post_status] => publish [comment_status] => open [ping_status] => open [post_password] => [post_name] => digital-product-design [to_ping] => [pinged] => [post_modified] => 2022-11-14 22:28:12 [post_modified_gmt] => 2022-11-15 03:28:12 [post_content_filtered] => [post_parent] => 0 [guid] => https://tech-clarity.com/?p=6963 [menu_order] => 0 [post_type] => post [post_mime_type] => [comment_count] => 0 [filter] => raw ) [3] => WP_Post Object ( [ID] => 6947 [post_author] => 2 [post_date] => 2018-05-29 19:09:13 [post_date_gmt] => 2018-05-29 23:09:13 [post_content] => [post_title] => PLM in the Cloud Webcast with Sogeti, and Microsoft
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => plm-cloud-webcast-sogeti-microsoft
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:46
[post_modified_gmt] => 2022-11-15 03:26:46
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6947
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[4] => WP_Post Object
(
[ID] => 6937
[post_author] => 2572
[post_date] => 2018-05-16 13:13:15
[post_date_gmt] => 2018-05-16 17:13:15
[post_content] => How can retail companies improve their decisions to survive in today's complex omnichannel environment and anticipate changes in trends in a dynamic and volatile market? Can predictive retail analytics help? If so, what should retailers look for in a solution?
[post_title] => PLM in the Cloud Webcast with Sogeti, and Microsoft
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => plm-cloud-webcast-sogeti-microsoft
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:46
[post_modified_gmt] => 2022-11-15 03:26:46
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6947
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[4] => WP_Post Object
(
[ID] => 6937
[post_author] => 2572
[post_date] => 2018-05-16 13:13:15
[post_date_gmt] => 2018-05-16 17:13:15
[post_content] => How can retail companies improve their decisions to survive in today's complex omnichannel environment and anticipate changes in trends in a dynamic and volatile market? Can predictive retail analytics help? If so, what should retailers look for in a solution?
Listen to Tech-Clarity's Michelle Boucher and PTC's Brad Thomas, Product Manager of the Retail Business Unit discuss predictive analytics for the retail industry. They cover topics such as:
- The value of connecting multiple databases on a single platform
- How applying machine learning to analytics helps retail companies
- Examples of how predictive analytics solutions can help retailers
 [post_title] => Retail Analytics Solutions (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => retail-analytics-solutions-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:57
[post_modified_gmt] => 2022-11-15 03:26:57
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6937
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[5] => WP_Post Object
(
[ID] => 6926
[post_author] => 2
[post_date] => 2018-05-11 14:00:31
[post_date_gmt] => 2018-05-11 18:00:31
[post_content] => How can manufacturers get more value from their digital initiatives by leveraging the Internet of Things (IoT)? Manufacturers who embrace digitalization gain increased agility, innovation, speed, quality, and profitability in design, development, manufacturing, and the service lifecycle. One of the key enablers of these improvements is establishing a closer connection between product models and how physical products operate in the field. The digital twin leverages detailed 3D models, IoT data, and analytics to significantly change the relationship manufacturers have with their products and their customers, driving new sources of value for both.
In this Engineering.com webinar sponsored by Siemens PLM, Tech-Clarity's Jim Brown will explore how companies that embrace digitalization and the digital twin will displace competitors who stick with the status quo.
[post_title] => Retail Analytics Solutions (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => retail-analytics-solutions-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:57
[post_modified_gmt] => 2022-11-15 03:26:57
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6937
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[5] => WP_Post Object
(
[ID] => 6926
[post_author] => 2
[post_date] => 2018-05-11 14:00:31
[post_date_gmt] => 2018-05-11 18:00:31
[post_content] => How can manufacturers get more value from their digital initiatives by leveraging the Internet of Things (IoT)? Manufacturers who embrace digitalization gain increased agility, innovation, speed, quality, and profitability in design, development, manufacturing, and the service lifecycle. One of the key enablers of these improvements is establishing a closer connection between product models and how physical products operate in the field. The digital twin leverages detailed 3D models, IoT data, and analytics to significantly change the relationship manufacturers have with their products and their customers, driving new sources of value for both.
In this Engineering.com webinar sponsored by Siemens PLM, Tech-Clarity's Jim Brown will explore how companies that embrace digitalization and the digital twin will displace competitors who stick with the status quo.
 Key takeaways include:
Key takeaways include:
- How a digital twin helps create competitive differentiation
- How manufacturers can transform product performance and service through better product insights
- How a digital innovation platform supports the digital twin
Listen to Claus Gärtner, Head of Operations & Quality IT at Roche Diabetes Care, René Zoelfl PTC Life Sciences Development Manager, and Tech-Clarity's Michelle Boucher discuss how to support the development of medical devices. This discussion was an interactive panel discussion highlighting topics such as:
- The PLM journey taken by Roche Diabetes Care
- What drove Roche Diabetes Care to adopt a PLM system
- The results Roche Diabetes Care have achieved with PLM
- Perspectives of software solution selection criteria to support quality initiatives.
 [post_title] => How Roche Diabetes Care Is Winning at PLM, Quality, and Compliance (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => roche_diabetes_plm_quality
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:51
[post_modified_gmt] => 2022-11-15 03:26:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6920
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[7] => WP_Post Object
(
[ID] => 6891
[post_author] => 2
[post_date] => 2018-04-10 10:44:34
[post_date_gmt] => 2018-04-10 14:44:34
[post_content] =>
[post_title] => How Roche Diabetes Care Is Winning at PLM, Quality, and Compliance (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => roche_diabetes_plm_quality
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:51
[post_modified_gmt] => 2022-11-15 03:26:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6920
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[7] => WP_Post Object
(
[ID] => 6891
[post_author] => 2
[post_date] => 2018-04-10 10:44:34
[post_date_gmt] => 2018-04-10 14:44:34
[post_content] =>  How can manufacturers quickly gain value from remote equipment monitoring using the Internet of Things to begin their digital and service transformations? The Monitoring Equipment with IoT Quick Start Guide eBook offers pragmatic advice for companies that want to jump-start their IoT initiative with a practical approach that leads to much more significant value over time.
Please enjoy the summary below.
For the full eBook click the title to download the full PDF (no charge, no registration required) thanks to our sponsor PTC.
How can manufacturers quickly gain value from remote equipment monitoring using the Internet of Things to begin their digital and service transformations? The Monitoring Equipment with IoT Quick Start Guide eBook offers pragmatic advice for companies that want to jump-start their IoT initiative with a practical approach that leads to much more significant value over time.
Please enjoy the summary below.
For the full eBook click the title to download the full PDF (no charge, no registration required) thanks to our sponsor PTC.
Jump-Start Your IoT Initiative
Manufacturers are making major business improvements using the Internet of Things (IoT). They’re delivering value to customers in new ways and exploring new business models like selling outcomes versus products or shifting to a predictive service model. Some companies are well on their way to this strategic value, while for others it may seem out of reach. How can your company jump-start your IoT initiative? One proven way to get started with IoT is by remotely monitoring machines, either in your own plants or in your customers’ facilities. This gives you the real-time information you need to quickly identify failures and fix equipment more efficiently, improving service while reducing cost. Remote equipment monitoring provides value in the short term and helps develop a strong analytics foundation for your broader IoT strategy. This Quick Start Guide shares suggestions on what companies can do to get equipment connected, start gathering data, and put the data to use. Let’s get started!
Next Steps
Remote monitoring using the IoT can provide rapid value by helping your company gain visibility to your equipment. The value is strategic, but getting started doesn’t need to be intimidating. You can start small and grow value over time. Your investment can provide a valuable foundation to build on as you connect more equipment or implement more advanced features.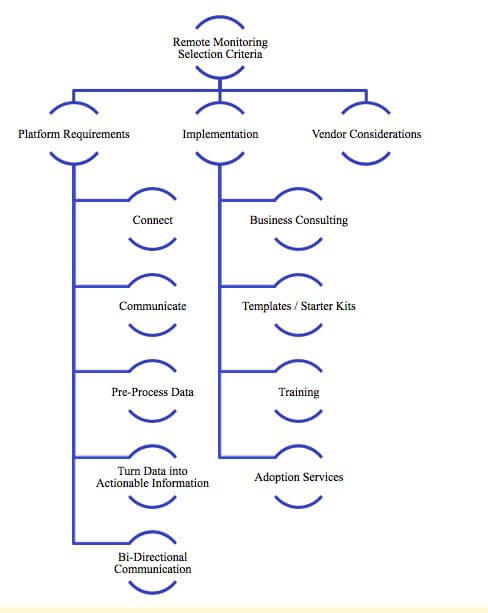 It’s important to find an IoT platform that offers a variety of predefined connectivity and application options so you don’t need to undertake a large integration project from scratch. Find a solution and IoT-savvy partner that can get you started with a simple project, but that offers the business knowledge and advanced tools needed to help you expand on your early success.
Remote equipment monitoring lets companies reduce service cost, increase service revenue, develop stronger relationships, and start the transition to proactive and predictive service. It’s time to get started to improve value for your company and your customers.
*This summary is an abbreviated version of the report and does not contain the full content. A link to download the full report is available above.
If you have difficulty obtaining a copy of the report, please contact us using the "Contact" link below.
[post_title] => IoT Machine Monitoring Quick Start Guide
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => iot-monitoring
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:27:55
[post_modified_gmt] => 2022-11-15 03:27:55
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6891
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[8] => WP_Post Object
(
[ID] => 6854
[post_author] => 2572
[post_date] => 2018-04-03 11:52:05
[post_date_gmt] => 2018-04-03 15:52:05
[post_content] =>
It’s important to find an IoT platform that offers a variety of predefined connectivity and application options so you don’t need to undertake a large integration project from scratch. Find a solution and IoT-savvy partner that can get you started with a simple project, but that offers the business knowledge and advanced tools needed to help you expand on your early success.
Remote equipment monitoring lets companies reduce service cost, increase service revenue, develop stronger relationships, and start the transition to proactive and predictive service. It’s time to get started to improve value for your company and your customers.
*This summary is an abbreviated version of the report and does not contain the full content. A link to download the full report is available above.
If you have difficulty obtaining a copy of the report, please contact us using the "Contact" link below.
[post_title] => IoT Machine Monitoring Quick Start Guide
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => iot-monitoring
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:27:55
[post_modified_gmt] => 2022-11-15 03:27:55
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6891
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[8] => WP_Post Object
(
[ID] => 6854
[post_author] => 2572
[post_date] => 2018-04-03 11:52:05
[post_date_gmt] => 2018-04-03 15:52:05
[post_content] =>  What steps should you take to transform your product design processes so you can develop smarter products?
Tech-Clarity's Top Five Tips to Transform Product Design for Smarter Products shares advice to help companies on their journey to develop smarter products. For many, making products smarter and connected is a key product trend that is becoming increasingly important for innovation and competitive differentiation. Plus many customers are starting to expect it. However, successfully developing smarter and connected products requires changes to your design process. At the same time, it's hard to know what you don't know. As you embark on that journey toward smarter and connected products, there will be lots of lessons learned. Based on survey research, Tech-Clarity has identified five tips to give you some hindsight and help you on that journey with the benefit of some of the lessons learned by other.
Please enjoy the summary* below.
For the full eBook, please visit our sponsor SOLIDWORKS (free of charge, registration required).
What steps should you take to transform your product design processes so you can develop smarter products?
Tech-Clarity's Top Five Tips to Transform Product Design for Smarter Products shares advice to help companies on their journey to develop smarter products. For many, making products smarter and connected is a key product trend that is becoming increasingly important for innovation and competitive differentiation. Plus many customers are starting to expect it. However, successfully developing smarter and connected products requires changes to your design process. At the same time, it's hard to know what you don't know. As you embark on that journey toward smarter and connected products, there will be lots of lessons learned. Based on survey research, Tech-Clarity has identified five tips to give you some hindsight and help you on that journey with the benefit of some of the lessons learned by other.
Please enjoy the summary* below.
For the full eBook, please visit our sponsor SOLIDWORKS (free of charge, registration required).
The Journey Toward New Development Approaches
 The emergence of smart, connected technologies is enabling unprecedented innovation. We have public trash cans that let trash collectors know when they need to be emptied. Pill bottles that remind you to take your medication. Complex machinery that notifies you when it needs servicing. The opportunities and growth potential seem limitless. Technology has become so critical, 52% of companies developing smart products fear losing market share unless they continually make products smarter. On top of that, 47% worry about reduced revenue.
With this in mind, we can expect to see a continual increase in the amount of software and electronics in products. Even 25% of companies that have not already made their products smarter, plan to add software and electronics over the next five years.
To successfully take advantage of the opportunity and transform products into intelligent systems, companies should embark on a journey to perfect development processes and manage the additional complexity. This eBook outlines steps to make your journey to transform your products a success.
The emergence of smart, connected technologies is enabling unprecedented innovation. We have public trash cans that let trash collectors know when they need to be emptied. Pill bottles that remind you to take your medication. Complex machinery that notifies you when it needs servicing. The opportunities and growth potential seem limitless. Technology has become so critical, 52% of companies developing smart products fear losing market share unless they continually make products smarter. On top of that, 47% worry about reduced revenue.
With this in mind, we can expect to see a continual increase in the amount of software and electronics in products. Even 25% of companies that have not already made their products smarter, plan to add software and electronics over the next five years.
To successfully take advantage of the opportunity and transform products into intelligent systems, companies should embark on a journey to perfect development processes and manage the additional complexity. This eBook outlines steps to make your journey to transform your products a success.
You Don’t Know What You Don’t Know
 While emerging technologies open doors for innovation and opportunity, they introduce new challenges. However, as you start your journey, it is hard to know what you don’t know. Learning from the experiences of others to minimize or avoid common challenges should be a first step. Even if you have already started your journey, knowing where to focus your energy for improvement can minimize risk. Based on the experiences of over 180 manufacturers, the top six challenges of making smarter products are:
While emerging technologies open doors for innovation and opportunity, they introduce new challenges. However, as you start your journey, it is hard to know what you don’t know. Learning from the experiences of others to minimize or avoid common challenges should be a first step. Even if you have already started your journey, knowing where to focus your energy for improvement can minimize risk. Based on the experiences of over 180 manufacturers, the top six challenges of making smarter products are:
- Products become much more complex
- Knowledge silos / lack of cross-functional knowledge
- Ensuring product requirements are met
- Lack of visibility into the impact of design decisions across disciplines
- Tools are incompatible across engineering disciplines
- Identifying system level problems sooner
 How can manufacturers design systems with greater confidence the system will work as designed?
Tech-Clarity's Smart Systems Buyer’s Guide for Systems Engineers and IoT Solution Architects helps manufacturers develop selection criteria to evaluate software solutions to support system design. The guide targets systems engineerings, IoT solution architects, and others involved with designing systems.
Tech-Clarity’s Buyer’s Guides go beyond software functionality to provide a framework of requirements that impact implementation success and long-term ROI.
Please enjoy the summary below, or click the report to download a PDF overview.
For the full report, please visit our sponsor, PTC, here (free of charge, registration required).
How can manufacturers design systems with greater confidence the system will work as designed?
Tech-Clarity's Smart Systems Buyer’s Guide for Systems Engineers and IoT Solution Architects helps manufacturers develop selection criteria to evaluate software solutions to support system design. The guide targets systems engineerings, IoT solution architects, and others involved with designing systems.
Tech-Clarity’s Buyer’s Guides go beyond software functionality to provide a framework of requirements that impact implementation success and long-term ROI.
Please enjoy the summary below, or click the report to download a PDF overview.
For the full report, please visit our sponsor, PTC, here (free of charge, registration required).
Table of Contents
- Executive Overview
- The Business Value of Systems Engineering
- Start with Process Definition
- Manage Requirements
- Design the System
- Design the System to Be Modular
- Support Product Line Variants
- Enable Detailed Design
- Support Connectivity
- Verify and Validate the System
- Assess Service Requirements
- Consider Vendor Attributes
- Identify Specific Needs for your Company
- Conclusion
- Recommendations
- About the Author
Executive Overview
In today’s world, fierce global competition is driving companies to seek new ways to competitively differentiate their products. Many companies are incorporating embedded software into their products to set their products apart from the competition. Embedded software offers opportunities to incorporate intelligence into a product as well as offer customers a more personalized experience. The Internet of Things (IoT) takes this even further by creating exciting possibilities for new and differentiated services for products with smart, connected devices. While bringing together mechanical components, electronics, and software presents exciting opportunities for innovation, it also brings unique challenges and adds new levels of complexity to today’s products and product development processes. The same goes for connected products as you add sensors, streaming data, and an ecosystem of connected systems. Whether you are a systems engineer or IoT solutions architect, expert systems engineering practices are critical to the success of today’s products. An essential part of implementing expert systems engineering practices is having the technology to support them. This buyer’s guide will help manufacturers select the right software to support systems engineering. This guide is composed of four major sections covering, systems engineering software tool functionality, service requirements, vendor attributes, and special company considerations (Figure 1). Each section includes a checklist of key requirements to investigate when selecting software tools to support systems engineering.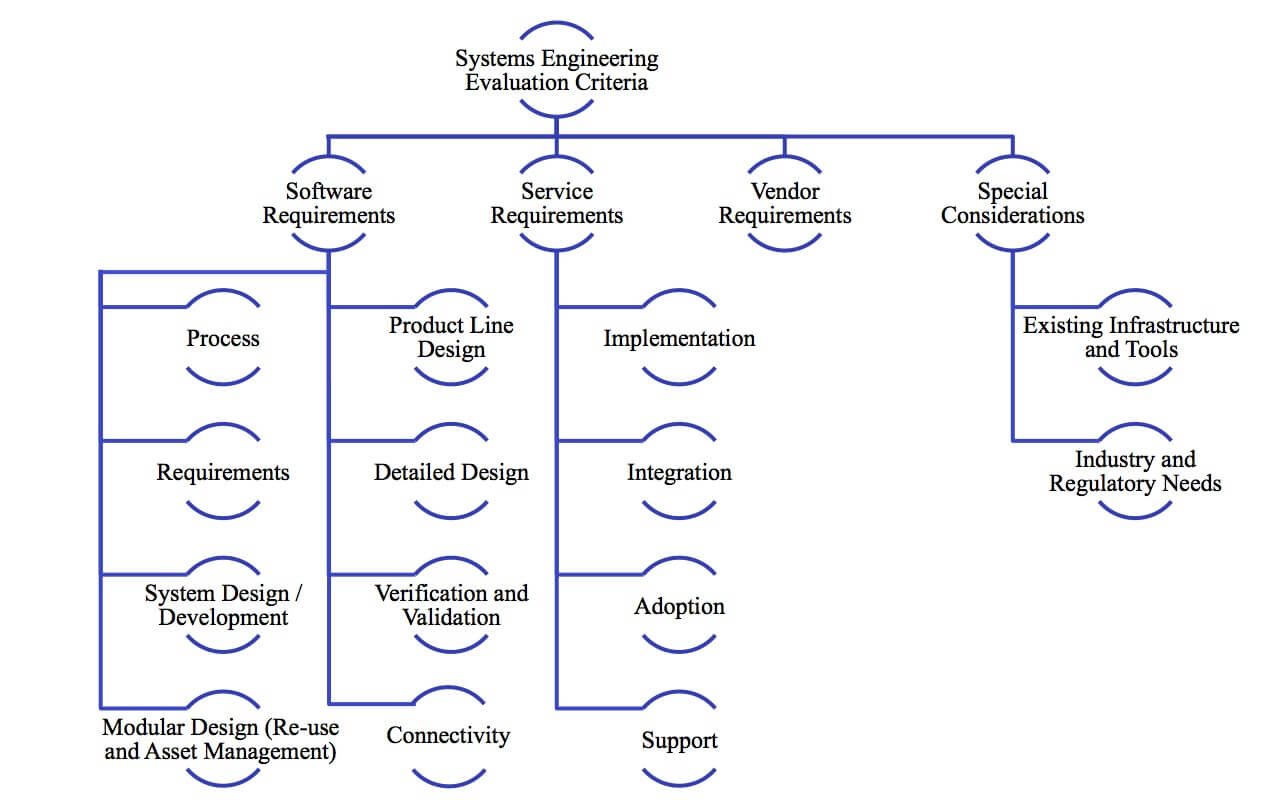 Figure 1: Systems Engineering Evaluation Framework
Figure 1: Systems Engineering Evaluation Framework
- IoT Solutions Architect
- DevOps IoT
- Internet of Things Solutions Engineer
- M2M and IoT Product Management and Engineering
- Technology Manager, Software Engineering (IoT)
- Solutions Engineer (IoT)
- Principal Technical Architect (IoT)
- Innovation Manager for Industry 4.0
- Product Manager
Conclusion
Expert systems engineering practices are key to taking advantage of innovation available through embedded software and the Internet of Things. The opportunities to create smart, connected devices can help companies set their products apart from the competition, helping them win new customers and increase revenues. However, bringing together systems of mechanical, electrical, and software components is complex. That complexity grows exponentially as companies try to meet the various needs of customers with different configurations. Connected systems add even further complexity as you add sensors, streaming data, and connected ecosystems. Complexity means there is risk things will go wrong. The impact of these problems can have a significant business impact and hurt the profitability of the product. Implementing expert systems engineering practices, with the right software tools to support them, can manage this complexity, making it easier to bring profitable products to market successfully. Even if today’s smart, connected devices are relatively simple, as they evolve and offer critical services such as those that impact safety, they will increase in complexity and need the same level of expert systems engineering practices. These practices and the supporting solution are not just limited to systems engineers either. There are a variety of IoT related roles involved with planning, designing, and architecting connected systems, such as IoT solution architects who will struggle with the exact same challenges as systems engineers. Companies planning for growth should consider both current and future needs. However, there are so many aspects of systems engineering; determining the right solution for your company can be very difficult. Using a high-level list of tool and process evaluation criteria such as the ones in this guide can help narrow down potential solutions by providing a quick “litmus test” to determine if a solution and partner are a good fit before conducting detailed functional or technical reviews. In the end, it is important to ensure that functionality, service, vendor, and special requirements are all considered when selecting a solution.Recommendations
Based on industry experience and research for this report, Tech-Clarity offers the following recommendations:- Identify and weigh systems engineering requirements based on company needs, existing applications, industry, and unique product and process requirements
- Use high-level requirements such as the ones in this guide to evaluate solutions based on business fit before engaging in detailed evaluations
- Consider long-term business and process growth needs and the potential to scale across product lines, departments, and engineering silos
- Consider all stages of systems engineering from process, requirements, design, and validation when investing in systems engineering solutions
- Think about all roles that can benefit from systems engineering best practices such as IoT solution architects
- Select a vendor who will be a trusted partner
 How can manufacturers ensure they get the most value from a new PDM solution? The PDM Buyer's Guide provides criteria for companies to select the right Production Data Management system to manage the complexity of today's products and product development environments. Manufacturers have many choices for managing critical CAD files and product data, including shared drives and the modern equivalents - DropBox, Drive, Google Drive, and a host of other options. But our research shows that they'll get more value from a structured, collaborative data management solution designed to support product development and further into the product lifecycle. This paper shares requirements for the basic capabilities companies need from PDM - Designing, Accessing, and Sharing product data.
Our Buyer's Guides look beyond software requirements to the other elements that help companies get value from their solutions, including criteria to consider for implementation, adoption, service, and vendor selection. This guide also covers special needs including advice for smaller and larger manufacturers and shares considerations for addressing some specific industry needs and supporting the transition to the digital enterprise.
Please enjoy the summary below, or click the report to download a PDF overview (no charge, no registration required).
For the full Buyer's Guide, please visit our sponsor PTC (no charge, registration required).
How can manufacturers ensure they get the most value from a new PDM solution? The PDM Buyer's Guide provides criteria for companies to select the right Production Data Management system to manage the complexity of today's products and product development environments. Manufacturers have many choices for managing critical CAD files and product data, including shared drives and the modern equivalents - DropBox, Drive, Google Drive, and a host of other options. But our research shows that they'll get more value from a structured, collaborative data management solution designed to support product development and further into the product lifecycle. This paper shares requirements for the basic capabilities companies need from PDM - Designing, Accessing, and Sharing product data.
Our Buyer's Guides look beyond software requirements to the other elements that help companies get value from their solutions, including criteria to consider for implementation, adoption, service, and vendor selection. This guide also covers special needs including advice for smaller and larger manufacturers and shares considerations for addressing some specific industry needs and supporting the transition to the digital enterprise.
Please enjoy the summary below, or click the report to download a PDF overview (no charge, no registration required).
For the full Buyer's Guide, please visit our sponsor PTC (no charge, registration required).
Table of Contents
- Introducing the PDM Buyer’s Guide
- The Product Data Management Imperative
- Analyze PDM Capabilities
- Assess Service Requirements
- Consider Vendor Requirements
- Identify Unique Company Needs
- Conclusion
- Recommendations
- About the Author
Introducing the PDM Buyer’s Guide
Product Data Management (PDM) is an important tool to help manufacturers overcome the complexities of designing, developing, producing, and supporting today’s products. Manual and ad-hoc approaches such as shared folders, FTP, Dropbox, box, and hard drives are simply not good solutions to manage critical, complex product information. These approaches may work for very small organizations, but quickly falter as organizations grow and must share information beyond a few core engineers. These techniques also fail to manage data relationships and complex file structures common to 3D CAD systems. PDM systems are purpose-built to address these issues. PDM is a structured, collaborative solution that helps manufacturers control, access, and share crucial product data. Selecting the right PDM system for your business has a large impact on productivity, product success, and profitability.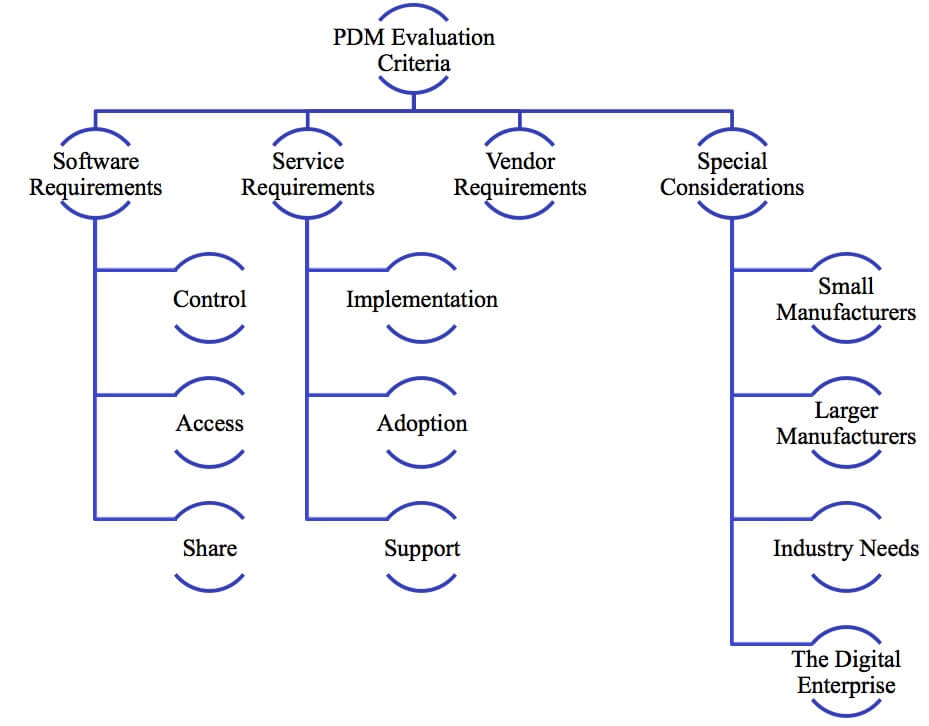 The PDM Buyer’s Guide is a reference tool to provide direction about what to look for when selecting a PDM system for your company. The guide is composed of four sections covering software functionality, service requirements, vendor attributes, and special company considerations. Each of these sections includes a checklist with key requirements to investigate when selecting PDM software. The guide focuses on common requirements that form the foundation of PDM for manufacturers:
The PDM Buyer’s Guide is a reference tool to provide direction about what to look for when selecting a PDM system for your company. The guide is composed of four sections covering software functionality, service requirements, vendor attributes, and special company considerations. Each of these sections includes a checklist with key requirements to investigate when selecting PDM software. The guide focuses on common requirements that form the foundation of PDM for manufacturers:
- Getting files under control so people can find the right revision with confidence
- Making sure concurrent updates don’t overwrite each other to avoid “the last save wins” syndrome
- Making information easily accessible and consumable to teams outside of Engineering for reviews and downstream processes
- Safely sharing information with customers, partners, and the supply chain
- Ensuring intellectual property (IP) is captured and securely accessible regardless of who stored it
- Getting away from complicated shared drive structures that lead to errors
- Making sure people don’t manufacture or purchase against the wrong drawing
- Providing “one version of the truth” versus multiple copies of designs
Conclusion
PDM helps manufacturers address market complexity and improve business performance. As Tech-Clarity’s Managing Engineering Data concludes, “Product data management is the fundamental building block of any engineering software strategy and helps companies get the most out of their precious engineering resources.” When evaluating PDM, manufacturers need to take into account: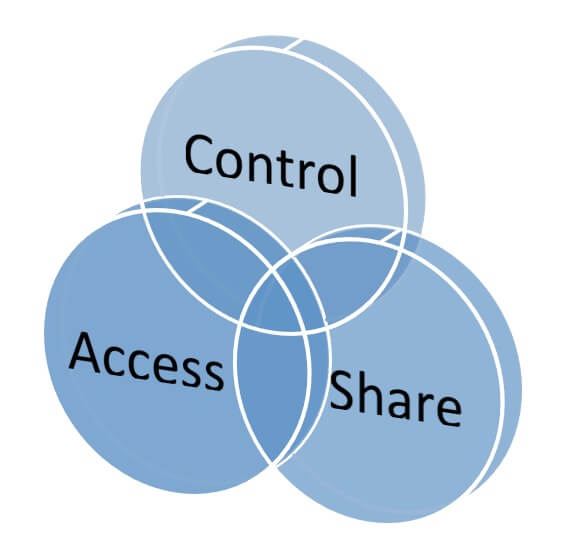 Product requirements
Product requirements- Implementation, adoption, and support requirements
- Vendor / business requirements
- Special requirements based on company size (particularly for very small or very large organizations)
- Special considerations to meet industry needs
- Future requirements as business continues to digitalize
Reccommendations
Based on industry experience and research for this report, Tech-Clarity offers the following recommendations:- Identify and weigh PDM requirements based on company needs, company size, industry, and any unique company needs
- Use high level requirements such as the ones in this guide to evaluate solutions based on business fit before engaging in detailed evaluations
- Consider using more simple, commodity technical solutions like cloud or managed services solutions for smaller companies, companies that wish to move quickly, or those with limited IT resources
- Take user adoption into account, including simplified access and increased visualization for non-engineering resources
- Consider using more full-featured, scalable solutions for larger organizations that have more demanding process and scalability needs and can afford the IT resources required to support capabilities such as site synchronization
- Take into account long-term business and process growth needs including digitalization and IoT initiatives
 How can monitoring equipment remotely provide value and help get your IoT initiative started? Jim Brown joins PTC's Michael Anderson for a webcast discussion to find out. Jim will share perspectives from his research including an overview of how remote equipment monitoring via the IoT can help drive significant service improvements. Jim will share tips from his upcoming Remote Equipment Monitoring Quick Start Guide including key things manufacturers should consider as they consider an initiative or select a solution to support this aspect of their digital transformation. Jim and Michael will also answer audience questions.
Register for the webinar now (no charge, registration required)
[post_title] => Jumpstarting IoT with Remote Equipment Monitoring (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => iot-monitoring-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:56
[post_modified_gmt] => 2022-11-15 03:26:56
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6775
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[12] => WP_Post Object
(
[ID] => 6742
[post_author] => 2572
[post_date] => 2018-03-08 11:30:30
[post_date_gmt] => 2018-03-08 16:30:30
[post_content] =>
How can monitoring equipment remotely provide value and help get your IoT initiative started? Jim Brown joins PTC's Michael Anderson for a webcast discussion to find out. Jim will share perspectives from his research including an overview of how remote equipment monitoring via the IoT can help drive significant service improvements. Jim will share tips from his upcoming Remote Equipment Monitoring Quick Start Guide including key things manufacturers should consider as they consider an initiative or select a solution to support this aspect of their digital transformation. Jim and Michael will also answer audience questions.
Register for the webinar now (no charge, registration required)
[post_title] => Jumpstarting IoT with Remote Equipment Monitoring (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => iot-monitoring-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:56
[post_modified_gmt] => 2022-11-15 03:26:56
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6775
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[12] => WP_Post Object
(
[ID] => 6742
[post_author] => 2572
[post_date] => 2018-03-08 11:30:30
[post_date_gmt] => 2018-03-08 16:30:30
[post_content] =>  What's the best way to make sure the product you design and build actually meets requirements? How do requirements management solutions help?
Listen and interview of SAFRAN Group's Yannick Laplume, Tech-Clarity's Michelle Boucher, and PTC's Christoph Braeuchle as they discuss the value of of a requirements management solution. They explore topics such as:
What's the best way to make sure the product you design and build actually meets requirements? How do requirements management solutions help?
Listen and interview of SAFRAN Group's Yannick Laplume, Tech-Clarity's Michelle Boucher, and PTC's Christoph Braeuchle as they discuss the value of of a requirements management solution. They explore topics such as:
- The business value of requirements management
- How to identify specific requirements needs
- How to select the right solution for your organization
 [post_title] => Requirements Management: How to select the right solution? (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => requirements-management-solution-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:56
[post_modified_gmt] => 2022-11-15 03:26:56
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6742
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[13] => WP_Post Object
(
[ID] => 6740
[post_author] => 2572
[post_date] => 2018-03-07 11:30:20
[post_date_gmt] => 2018-03-07 16:30:20
[post_content] =>
[post_title] => Requirements Management: How to select the right solution? (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => requirements-management-solution-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:56
[post_modified_gmt] => 2022-11-15 03:26:56
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6742
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[13] => WP_Post Object
(
[ID] => 6740
[post_author] => 2572
[post_date] => 2018-03-07 11:30:20
[post_date_gmt] => 2018-03-07 16:30:20
[post_content] =>  Does having multiple systems to manage product information create challenges for your company? Do you find some systems are so heavily customized, it's hard to take advantage of the latest software enhancements? How can a PLM medical device solution help? If you have thought about any of these questions, you will find this webinar especially interesting, especially if you are in the medical device industry.
Does having multiple systems to manage product information create challenges for your company? Do you find some systems are so heavily customized, it's hard to take advantage of the latest software enhancements? How can a PLM medical device solution help? If you have thought about any of these questions, you will find this webinar especially interesting, especially if you are in the medical device industry.
Listen to Tina Kunshier of Boston Scientific and Tech-Clarity's Michelle Boucher discuss how to select the right software solution to support the development of medical devices. This discussion will be a live interview highlighting topics such as:
- The PLM journey taken by Boston Scientific
- What drove Boston Scientific to consolidate PLM systems
- How Boston Scientific executed their strategy for a unified PLM system
- Selection criteria you should look for in a PLM solution, especially for medical device companies
- Advice to improve product development efficiencies and support regulatory compliance
 [post_title] => How Boston Scientific Selected their PLM Medical Device Software Solution (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => boston-scientific-plm-medical-device
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6740
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[14] => WP_Post Object
(
[ID] => 6732
[post_author] => 2572
[post_date] => 2018-03-05 11:56:22
[post_date_gmt] => 2018-03-05 16:56:22
[post_content] =>
[post_title] => How Boston Scientific Selected their PLM Medical Device Software Solution (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => boston-scientific-plm-medical-device
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6740
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[14] => WP_Post Object
(
[ID] => 6732
[post_author] => 2572
[post_date] => 2018-03-05 11:56:22
[post_date_gmt] => 2018-03-05 16:56:22
[post_content] => Listen to, PTC's Dave Duncan and Tech-Clarity's Michelle Boucher discuss how to improve the accuracy of service information for field personnel. They share stories about Embraer, Airbus Helicopters, and Kirloskar Oil Engines and explain how they are transforming their service organizations. During this webcast, you will also learn:
- How poor service information affects technician productivity, customer satisfaction, and after-sales operations
- Best practices for producing accurate service documentation based on Tech-Clarity research
- What you can do to keep service manuals up-to-date so that field technicians can trust service documentation
- Tips to support the cultural changes required to support the transformation for a more service oriented company
 [post_title] => Service Information: The Pivotal Factor in Your Aftermarket Business (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => service-information-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6732
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[15] => WP_Post Object
(
[ID] => 6721
[post_author] => 2
[post_date] => 2018-02-28 08:48:20
[post_date_gmt] => 2018-02-28 13:48:20
[post_content] => How can digitalization help medical device companies improve innovation and drive profitable growth despite increased complexity and regulatory scrutiny? This animated video offers a look at what a digital medical device company looks like and offers three initiatives companies can adopt to start their digital transformation.
Digitalization offers significant benefits to medical device manufacturers because the status quo is no longer good enough. It relies too much on paper and electronic documents, inevitability leading to missed deadlines, mistakes, and inefficiency. In today's environment, key contributors work independently on their own information, leading to integration errors, compliance issues, and market delays. Now, companies can transform their businesses so that each step in the development process leverages digital information from the prior step, takes advantage of comprehensive 3D product models, and creates a complete digital thread. Digitalization can then extend into manufacturing and enable device manufacturers to quickly commission production and track manufacturing.
Medical Device companies have the opportunity to leverage digitalization and the digital enterprise to develop innovative products and bring them to market quickly. By digitalizing designs and workflows, digital medical device companies can:
[post_title] => Service Information: The Pivotal Factor in Your Aftermarket Business (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => service-information-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6732
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[15] => WP_Post Object
(
[ID] => 6721
[post_author] => 2
[post_date] => 2018-02-28 08:48:20
[post_date_gmt] => 2018-02-28 13:48:20
[post_content] => How can digitalization help medical device companies improve innovation and drive profitable growth despite increased complexity and regulatory scrutiny? This animated video offers a look at what a digital medical device company looks like and offers three initiatives companies can adopt to start their digital transformation.
Digitalization offers significant benefits to medical device manufacturers because the status quo is no longer good enough. It relies too much on paper and electronic documents, inevitability leading to missed deadlines, mistakes, and inefficiency. In today's environment, key contributors work independently on their own information, leading to integration errors, compliance issues, and market delays. Now, companies can transform their businesses so that each step in the development process leverages digital information from the prior step, takes advantage of comprehensive 3D product models, and creates a complete digital thread. Digitalization can then extend into manufacturing and enable device manufacturers to quickly commission production and track manufacturing.
Medical Device companies have the opportunity to leverage digitalization and the digital enterprise to develop innovative products and bring them to market quickly. By digitalizing designs and workflows, digital medical device companies can:
- Connect data and processes across disciplines and the lifecycle
- Leverage digital data from one step to the next
- Streamline engineering
- Reduce errors
- Improve traceability
- Digital design for medical device companies (overview)
- Digital design
- Digital design transfer
- Paperless manufacturing
 What should manufacturers look for when they buy cloud software for Product Lifecycle Management? Our PLM Cloud Buyer's Guide shares the important factors companies need to know when they evaluate cloud solutions. The guide explains the reasons companies are pursuing cloud options including standard benefits like reduced implementation cost and faster time to benefit, but also highlights specific advantages to product developers including the ability to better support global environments and to design-anywhere, build-anywhere. The eBook also shares the benefits for IT departments such as the ability to provide cloud-level performance and offload operational overhead to focus on more value-added activities. But despite the benefits of the cloud, the guide explains the need to have a fully capable PLM system as the highest priority before prioritizing the deployment option, what we call a solution-first approach.
As with our other Buyer's Guides, the guide goes beyond software requirements to share deployment options, pricing considerations, implementation factors, vendor considerations, and other special requirements such as industry needs. The guide also points out the need to select a solution and a partner that will serve as the foundation for continued growth as PLM continues to expand as the digital innovation backbone and serves the future needs of the digital enterprise with IoT, AR, the digital twin, and more.
Please enjoy the summary below, or click the report to download a PDF overview (free of charge, no registration required).
For the full Buyer's Guide, please visit our sponsor PTC.
What should manufacturers look for when they buy cloud software for Product Lifecycle Management? Our PLM Cloud Buyer's Guide shares the important factors companies need to know when they evaluate cloud solutions. The guide explains the reasons companies are pursuing cloud options including standard benefits like reduced implementation cost and faster time to benefit, but also highlights specific advantages to product developers including the ability to better support global environments and to design-anywhere, build-anywhere. The eBook also shares the benefits for IT departments such as the ability to provide cloud-level performance and offload operational overhead to focus on more value-added activities. But despite the benefits of the cloud, the guide explains the need to have a fully capable PLM system as the highest priority before prioritizing the deployment option, what we call a solution-first approach.
As with our other Buyer's Guides, the guide goes beyond software requirements to share deployment options, pricing considerations, implementation factors, vendor considerations, and other special requirements such as industry needs. The guide also points out the need to select a solution and a partner that will serve as the foundation for continued growth as PLM continues to expand as the digital innovation backbone and serves the future needs of the digital enterprise with IoT, AR, the digital twin, and more.
Please enjoy the summary below, or click the report to download a PDF overview (free of charge, no registration required).
For the full Buyer's Guide, please visit our sponsor PTC.
Cloud PLM Adoption and Buyer's Guide Introduction
Although manufacturers have started to adopt cloud solutions for many aspects of their business, Product Lifecycle Management (PLM) has lagged behind. The transition, however, is picking up pace. More companies are considering cloud PLM and many already leverage the cloud to get more value from PLM (and achieve that value faster). Given the increased interest and adoption, it’s time to put together some selection criteria to help companies with their decisions. We recommend that companies pick their solution first and then choose their deployment option. This is effectively a “solution first” approach as opposed to a “cloud first” approach. We find that companies are just not willing to shortchange functionality in this crucial area. They recognize it’s important to evaluate the functional capabilities of a PLM system to ensure they’ll gain the significant top- and bottom-line benefits that PLM delivers.
There are still important decisions to make after a solution is selected. Many systems can be deployed in a variety of ways ranging from cloud Software as a Service (SaaS) to traditional, on-premise implementations. The deployment choice impacts important factors including cost, security, resource requirements, performance, risk, and time to benefit. This guide is designed to help companies navigate the options and choose the best-suited PLM cloud option for their business.
*This summary is an abbreviated version of the report and does not contain the full content. A link to download the full report is available above.
[post_title] => Cloud PLM Buyer's Guide
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => cloud-plm-guide
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:27:54
[post_modified_gmt] => 2022-11-15 03:27:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6708
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[17] => WP_Post Object
(
[ID] => 6700
[post_author] => 2
[post_date] => 2018-02-20 11:20:02
[post_date_gmt] => 2018-02-20 16:20:02
[post_content] => Medical Device companies have to manage complexity and regulation without placing excess overhead on their designers. Devices complexity has led to increased complaints, CAPAs, observations, and recalls. The root cause is frequently insufficient and cumbersome design control.
Digitalization helps them design with agility while maintaining control. It improves speed, accuracy, and productivity by automating submissions, DMRs, and DHFs from design data. The digital approach transitions device manufacturers away from document-centric and paper-based processes, or the electronic equivalent of processes that originated on paper that have been automated but never digitalized.
Digital design leverages complete digital models and creates a complete digital thread of information through design and the product lifecycle. It allows a cohesive approach where each step in design builds on the previous, and results in a digital model that engineers can use to validate and verify the behavior of the physical product in digital form using simulation.
Digital design control helps medical device companies get devices approved more quickly, keeps quality high, reduces risk, controls cost, and helps companies get innovative product to market quickly. The results are faster time to market, increased quality, and improved compliance.
This video series is sponsored by Siemens, a leader in digitalization for the manufacturing industries.
Click here for more information on intelligent design control in the medical device industry from our sponsor, Siemens, a leader in digitalization for the manufacturing industries.
https://youtu.be/z2zyO0O5o_Q
[post_title] => Digital Design Control for Medical Devices
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => med-dev-control
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:28:47
[post_modified_gmt] => 2022-11-15 03:28:47
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6700
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[18] => WP_Post Object
(
[ID] => 6645
[post_author] => 2
[post_date] => 2018-02-07 06:48:42
[post_date_gmt] => 2018-02-07 11:48:42
[post_content] => Medical Device manufacturers struggling with product complexity can leverage digitalization to more efficiently transfer product designs to the shop floor. This episode of Tech-Clarity TV shares how digitalization can combat long validation times and slow manufacturing ramp-up by creating a digital continuity between product designs and manufacturing processes. Digital design transfer brings the potential for:
Given the increased interest and adoption, it’s time to put together some selection criteria to help companies with their decisions. We recommend that companies pick their solution first and then choose their deployment option. This is effectively a “solution first” approach as opposed to a “cloud first” approach. We find that companies are just not willing to shortchange functionality in this crucial area. They recognize it’s important to evaluate the functional capabilities of a PLM system to ensure they’ll gain the significant top- and bottom-line benefits that PLM delivers.
There are still important decisions to make after a solution is selected. Many systems can be deployed in a variety of ways ranging from cloud Software as a Service (SaaS) to traditional, on-premise implementations. The deployment choice impacts important factors including cost, security, resource requirements, performance, risk, and time to benefit. This guide is designed to help companies navigate the options and choose the best-suited PLM cloud option for their business.
*This summary is an abbreviated version of the report and does not contain the full content. A link to download the full report is available above.
[post_title] => Cloud PLM Buyer's Guide
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => cloud-plm-guide
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:27:54
[post_modified_gmt] => 2022-11-15 03:27:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6708
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[17] => WP_Post Object
(
[ID] => 6700
[post_author] => 2
[post_date] => 2018-02-20 11:20:02
[post_date_gmt] => 2018-02-20 16:20:02
[post_content] => Medical Device companies have to manage complexity and regulation without placing excess overhead on their designers. Devices complexity has led to increased complaints, CAPAs, observations, and recalls. The root cause is frequently insufficient and cumbersome design control.
Digitalization helps them design with agility while maintaining control. It improves speed, accuracy, and productivity by automating submissions, DMRs, and DHFs from design data. The digital approach transitions device manufacturers away from document-centric and paper-based processes, or the electronic equivalent of processes that originated on paper that have been automated but never digitalized.
Digital design leverages complete digital models and creates a complete digital thread of information through design and the product lifecycle. It allows a cohesive approach where each step in design builds on the previous, and results in a digital model that engineers can use to validate and verify the behavior of the physical product in digital form using simulation.
Digital design control helps medical device companies get devices approved more quickly, keeps quality high, reduces risk, controls cost, and helps companies get innovative product to market quickly. The results are faster time to market, increased quality, and improved compliance.
This video series is sponsored by Siemens, a leader in digitalization for the manufacturing industries.
Click here for more information on intelligent design control in the medical device industry from our sponsor, Siemens, a leader in digitalization for the manufacturing industries.
https://youtu.be/z2zyO0O5o_Q
[post_title] => Digital Design Control for Medical Devices
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => med-dev-control
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:28:47
[post_modified_gmt] => 2022-11-15 03:28:47
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6700
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[18] => WP_Post Object
(
[ID] => 6645
[post_author] => 2
[post_date] => 2018-02-07 06:48:42
[post_date_gmt] => 2018-02-07 11:48:42
[post_content] => Medical Device manufacturers struggling with product complexity can leverage digitalization to more efficiently transfer product designs to the shop floor. This episode of Tech-Clarity TV shares how digitalization can combat long validation times and slow manufacturing ramp-up by creating a digital continuity between product designs and manufacturing processes. Digital design transfer brings the potential for:
- Simulating product, processes, lines, and plants
- Virtually commissioning production equipment
- Creating a seamless digital thread of information
- Transferring production process data to MES systems
 Jim Brown joins PTC's Alan Goldman and Mark Lobo for a web panel discussion on adopting Cloud PLM. Jim will share perspectives from his recent research including cloud PLM benefits and requirements. Jim will share insights from his upcoming Cloud PLM Buyer's guide including criteria companies should look for in a solution, for adoption, and their vendor partner. Jim and the other presenters will also anwer audience questions.
Jim, Alan, and Mark will be joined by Tim Curran of KPIT for a North American panel on Engineering360 on the same topic.
Register for the European webcast now (replay pending) (free of charge, registration required).
Register for the North American webinar now (free of charge, registration required)
[post_title] => Selecting the Right Cloud PLM System (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => cloud-plm-3
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => http://tech-clarity.com/?p=6633
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
)
[post_count] => 20
[current_post] => -1
[before_loop] => 1
[in_the_loop] =>
[post] => WP_Post Object
(
[ID] => 6989
[post_author] => 2
[post_date] => 2018-06-18 14:39:59
[post_date_gmt] => 2018-06-18 18:39:59
[post_content] =>
Jim Brown joins PTC's Alan Goldman and Mark Lobo for a web panel discussion on adopting Cloud PLM. Jim will share perspectives from his recent research including cloud PLM benefits and requirements. Jim will share insights from his upcoming Cloud PLM Buyer's guide including criteria companies should look for in a solution, for adoption, and their vendor partner. Jim and the other presenters will also anwer audience questions.
Jim, Alan, and Mark will be joined by Tim Curran of KPIT for a North American panel on Engineering360 on the same topic.
Register for the European webcast now (replay pending) (free of charge, registration required).
Register for the North American webinar now (free of charge, registration required)
[post_title] => Selecting the Right Cloud PLM System (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => cloud-plm-3
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:54
[post_modified_gmt] => 2022-11-15 03:26:54
[post_content_filtered] =>
[post_parent] => 0
[guid] => http://tech-clarity.com/?p=6633
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
)
[post_count] => 20
[current_post] => -1
[before_loop] => 1
[in_the_loop] =>
[post] => WP_Post Object
(
[ID] => 6989
[post_author] => 2
[post_date] => 2018-06-18 14:39:59
[post_date_gmt] => 2018-06-18 18:39:59
[post_content] =>  How do top performing companies plan and deliver profitable portfolios of smarter, more connected products? Join this webcast to hear what recent survey results tell us about developing innovative products in the digital age. The webcast includes results from the 6th Product Portfolio Management Benchmark study, conducted by Tech-Clarity and sponsored by Planview.
Hear Tech-Clarity's Jim Brown and Planview's Carrie Nauyalis share their insights about best practices to find out some surprising things that companies that deliver more innovative, profitable product portfolios than their competitors do differently.
Register for the Planview sponsored webcast now (no charge, registration required).
[post_title] => Secrets to Delivering Smart, Connected Product Portfolios (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => smart-portfolios-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:57
[post_modified_gmt] => 2022-11-15 03:26:57
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6989
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[comment_count] => 0
[current_comment] => -1
[found_posts] => 895
[max_num_pages] => 45
[max_num_comment_pages] => 0
[is_single] =>
[is_preview] =>
[is_page] =>
[is_archive] =>
[is_date] =>
[is_year] =>
[is_month] =>
[is_day] =>
[is_time] =>
[is_author] =>
[is_category] =>
[is_tag] =>
[is_tax] =>
[is_search] =>
[is_feed] =>
[is_comment_feed] =>
[is_trackback] =>
[is_home] => 1
[is_privacy_policy] =>
[is_404] =>
[is_embed] =>
[is_paged] =>
[is_admin] =>
[is_attachment] =>
[is_singular] =>
[is_robots] =>
[is_favicon] =>
[is_posts_page] =>
[is_post_type_archive] =>
[query_vars_hash:WP_Query:private] => 3b6dea43c30c452b522477188fac6b25
[query_vars_changed:WP_Query:private] => 1
[thumbnails_cached] =>
[allow_query_attachment_by_filename:protected] =>
[stopwords:WP_Query:private] =>
[compat_fields:WP_Query:private] => Array
(
[0] => query_vars_hash
[1] => query_vars_changed
)
[compat_methods:WP_Query:private] => Array
(
[0] => init_query_flags
[1] => parse_tax_query
)
[query_cache_key:WP_Query:private] => wp_query:fdbaf4e574486e9122ec25c6377c060d:0.92606000 17796061100.93591700 1779606110
)
How do top performing companies plan and deliver profitable portfolios of smarter, more connected products? Join this webcast to hear what recent survey results tell us about developing innovative products in the digital age. The webcast includes results from the 6th Product Portfolio Management Benchmark study, conducted by Tech-Clarity and sponsored by Planview.
Hear Tech-Clarity's Jim Brown and Planview's Carrie Nauyalis share their insights about best practices to find out some surprising things that companies that deliver more innovative, profitable product portfolios than their competitors do differently.
Register for the Planview sponsored webcast now (no charge, registration required).
[post_title] => Secrets to Delivering Smart, Connected Product Portfolios (webcast)
[post_excerpt] =>
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => smart-portfolios-webcast
[to_ping] =>
[pinged] =>
[post_modified] => 2022-11-14 22:26:57
[post_modified_gmt] => 2022-11-15 03:26:57
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://tech-clarity.com/?p=6989
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[comment_count] => 0
[current_comment] => -1
[found_posts] => 895
[max_num_pages] => 45
[max_num_comment_pages] => 0
[is_single] =>
[is_preview] =>
[is_page] =>
[is_archive] =>
[is_date] =>
[is_year] =>
[is_month] =>
[is_day] =>
[is_time] =>
[is_author] =>
[is_category] =>
[is_tag] =>
[is_tax] =>
[is_search] =>
[is_feed] =>
[is_comment_feed] =>
[is_trackback] =>
[is_home] => 1
[is_privacy_policy] =>
[is_404] =>
[is_embed] =>
[is_paged] =>
[is_admin] =>
[is_attachment] =>
[is_singular] =>
[is_robots] =>
[is_favicon] =>
[is_posts_page] =>
[is_post_type_archive] =>
[query_vars_hash:WP_Query:private] => 3b6dea43c30c452b522477188fac6b25
[query_vars_changed:WP_Query:private] => 1
[thumbnails_cached] =>
[allow_query_attachment_by_filename:protected] =>
[stopwords:WP_Query:private] =>
[compat_fields:WP_Query:private] => Array
(
[0] => query_vars_hash
[1] => query_vars_changed
)
[compat_methods:WP_Query:private] => Array
(
[0] => init_query_flags
[1] => parse_tax_query
)
[query_cache_key:WP_Query:private] => wp_query:fdbaf4e574486e9122ec25c6377c060d:0.92606000 17796061100.93591700 1779606110
)
All Results for "All"
Bridging the Gap Between Engineering and Purchasing
How can manufacturers bridge the knowledge gap between design and procurement? The Manufacturer’s Guide to Bridging the Engineering-Purchasing Gap shares our perspective on how manufacturers can take a digital approach to sharing critical BOM data from Engineering with Purchasing so they can reduce cost, improve efficiency, and prevent late shipments due to part shortages. Please…
Guide to Selecting a Smart, Connected Product Design Solution (eBook)
How does product design, development, and engineering need to change in order to support digital products? Key Considerations for Selecting a Smart, Connected Product Design Solution eBook shares that today’s new product development (NPD) processes simply aren’t comprehensive enough to effectively design smart, connected products. What do manufacturers need to look for in their engineering…
PLM in the Cloud Webcast with Sogeti, and Microsoft
PLM helps manufacturers improve innovation and product development performance through streamlined processes, centralized data, and integrated tools. It’s a key part of any manufacturer’s digitalization strategy. Learn how manufacturers are leveraging the cloud to remove the barriers to achieving the strategic benefits of Product Lifecycle Management. Join an interesting panel discussion between Tech-Clarity, Sogeti, and…
Retail Analytics Solutions (webcast)
How can retail companies improve their decisions to survive in today’s complex omnichannel environment and anticipate changes in trends in a dynamic and volatile market? Can predictive retail analytics help? If so, what should retailers look for in a solution? Listen to Tech-Clarity’s Michelle Boucher and PTC’s Brad Thomas, Product Manager of the Retail Business…
Enabling the Digital Twin with IoT (webcast)
How can manufacturers get more value from their digital initiatives by leveraging the Internet of Things (IoT)? Manufacturers who embrace digitalization gain increased agility, innovation, speed, quality, and profitability in design, development, manufacturing, and the service lifecycle. One of the key enablers of these improvements is establishing a closer connection between product models and how…
How Roche Diabetes Care Is Winning at PLM, Quality, and Compliance (webcast)
How can medical device companies balance quality, innovation, compliance, and profitability? Where does a PLM solution fit in? Is it possible to achieve a single source of truth? Listen to Claus Gärtner, Head of Operations & Quality IT at Roche Diabetes Care, René Zoelfl PTC Life Sciences Development Manager, and Tech-Clarity’s Michelle Boucher discuss how to support the…
IoT Machine Monitoring Quick Start Guide
How can manufacturers quickly gain value from remote equipment monitoring using the Internet of Things to begin their digital and service transformations? The Monitoring Equipment with IoT Quick Start Guide eBook offers pragmatic advice for companies that want to jump-start their IoT initiative with a practical approach that leads to much more significant value over…
Top 5 Tips to Transforming Product Design for Smarter Products
What steps should you take to transform your product design processes so you can develop smarter products? Tech-Clarity’s Top Five Tips to Transform Product Design for Smarter Products shares advice to help companies on their journey to develop smarter products. For many, making products smarter and connected is a key product trend that is becoming…
Smart Systems Buyer’s Guide for Systems Engineers and IoT Solution Architects (white paper)
How can manufacturers design systems with greater confidence the system will work as designed? Tech-Clarity’s Smart Systems Buyer’s Guide for Systems Engineers and IoT Solution Architects helps manufacturers develop selection criteria to evaluate software solutions to support system design. The guide targets systems engineerings, IoT solution architects, and others involved with designing systems. Tech-Clarity’s Buyer’s Guides…
Product Data Management Buyer’s Guide (white paper)
How can manufacturers ensure they get the most value from a new PDM solution? The PDM Buyer’s Guide provides criteria for companies to select the right Production Data Management system to manage the complexity of today’s products and product development environments. Manufacturers have many choices for managing critical CAD files and product data, including shared…
Jumpstarting IoT with Remote Equipment Monitoring (webcast)
How can monitoring equipment remotely provide value and help get your IoT initiative started? Jim Brown joins PTC’s Michael Anderson for a webcast discussion to find out. Jim will share perspectives from his research including an overview of how remote equipment monitoring via the IoT can help drive significant service improvements. Jim will share tips from his upcoming Remote…
Requirements Management: How to select the right solution? (webcast)
What’s the best way to make sure the product you design and build actually meets requirements? How do requirements management solutions help? Listen and interview of SAFRAN Group‘s Yannick Laplume, Tech-Clarity’s Michelle Boucher, and PTC’s Christoph Braeuchle as they discuss the value of of a requirements management solution. They explore topics such as: The business value of requirements management How…
How Boston Scientific Selected their PLM Medical Device Software Solution (webcast)
Does having multiple systems to manage product information create challenges for your company? Do you find some systems are so heavily customized, it’s hard to take advantage of the latest software enhancements? How can a PLM medical device solution help? If you have thought about any of these questions, you will find this webinar especially…
Service Information: The Pivotal Factor in Your Aftermarket Business (webcast)
Listen to, PTC’s Dave Duncan and Tech-Clarity’s Michelle Boucher discuss how to improve the accuracy of service information for field personnel. They share stories about Embraer, Airbus Helicopters, and Kirloskar Oil Engines and explain how they are transforming their service organizations. During this webcast, you will also learn: How poor service information affects technician productivity, customer satisfaction,…
Digitalization in the Medical Device Industry (animation)
How can digitalization help medical device companies improve innovation and drive profitable growth despite increased complexity and regulatory scrutiny? This animated video offers a look at what a digital medical device company looks like and offers three initiatives companies can adopt to start their digital transformation. Digitalization offers significant benefits to medical device manufacturers because…
Cloud PLM Buyer’s Guide
What should manufacturers look for when they buy cloud software for Product Lifecycle Management? Our PLM Cloud Buyer’s Guide shares the important factors companies need to know when they evaluate cloud solutions. The guide explains the reasons companies are pursuing cloud options including standard benefits like reduced implementation cost and faster time to benefit, but…
Digital Design Control for Medical Devices
Medical Device companies have to manage complexity and regulation without placing excess overhead on their designers. Devices complexity has led to increased complaints, CAPAs, observations, and recalls. The root cause is frequently insufficient and cumbersome design control. Digitalization helps them design with agility while maintaining control. It improves speed, accuracy, and productivity by automating submissions,…
Digital Design Transfer for Medical Devices (video)
Medical Device manufacturers struggling with product complexity can leverage digitalization to more efficiently transfer product designs to the shop floor. This episode of Tech-Clarity TV shares how digitalization can combat long validation times and slow manufacturing ramp-up by creating a digital continuity between product designs and manufacturing processes. Digital design transfer brings the potential for:…
Selecting the Right Cloud PLM System (webcast)
Jim Brown joins PTC’s Alan Goldman and Mark Lobo for a web panel discussion on adopting Cloud PLM. Jim will share perspectives from his recent research including cloud PLM benefits and requirements. Jim will share insights from his upcoming Cloud PLM Buyer’s guide including criteria companies should look for in a solution, for adoption, and their vendor partner….






















